Our last blog post about Chernobyl generated a lot of interest among our friends, but seemed to raise more questions than it answered. Many people asked for more basic information about radiation and more comparisons between Chernobyl and Japan, and safe levels versus dangerous levels. In light of that, we have compiled this follow-up post.
First Things First – What is Radiation (in plain English)?
In our last post, we showed lots of pictures of this little yellow Geiger counter that we used to measure radiation. So what is this thing measuring?

Basically, it’s measuring both particles and waves in the air around it. Some radiation is transported via particles that can easily be blocked by a piece of paper, or a jacket. Other types of radiation exist as waves, that can move straight through a wall. This devise is measuring both (two types of particles and one wave) and it’s adding them up to give us a total level of radiation.
The level of radiation is expressed as an amount of radiation (in a unit called Sieverts) per hour of exposure. So if the Geiger counter reads 0.22 microSieverts per hour (as it does in the photo above), that means I received .22 microSieverts of radiation while having my hour-long breakfast in Kiev.
The New York Times has a nice article providing a bit more detail about these three types of radiation and other terms commonly associated with radiation.
How Much Radiation Is Dangerous?
It depends who you ask. Some people will say no amount is safe, but that doesn’t really help us understand the relative dangers. Here are some basic numbers to use as a guide (μSv means microSieverts):
10 μSv – The average radiation you received today
40 μSv – The radiation you receive by taking a flight from New York to L.A.
100 μSv – The radiation you receive during a dental x-ray
800 μSv – Total radiation dose at Three-Mile Island for the duration of the accident
3,000 μSv – Radiation dose from a mammogram
3,600 μSv – Average radiation a US citizen receives in a year from all sources
50,000 μSv – Maximum allowable yearly occupational dose (USA)
100,000 μSv – Lowest yearly dose likely linked to increased cancer risk
2,000,000 μSv – Severe radiation poisoning (sometimes fatal)
So How Dangerous Are The Levels In Japan?
The short answer is, not very, unless you are close to the Fukushima plant. Keeping the standards described above in mind, let’s examine this map of radiation levels in Japan.
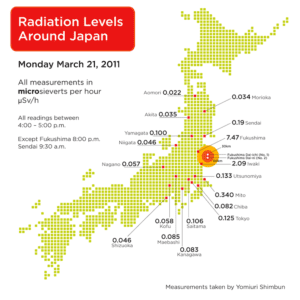
Tokyo usually has a background radiation level of .04 μSv/h. On April 5, the level was .12 μSv/h. That’s more than double, but is it dangerous? The short answer is no. If we multiply .12 by the number of hours in a year (about 8766) we see that a Tokyo resident would receive about 1,052 μSv of radiation a year at current levels. That is about 2% of the amount that the USA allows workers to receive on the job, or about a third of the amount the average American receives per year from all sources.
This is not meant to downplay that risks at the Japanese plant. As Chernobyl showed us, this is a technology that can quickly escape our control and cause massive devastation. However, while many areas in japan are experiencing radiation levels above the normal background level, the levels in most place are not likely dangerous.
What About Near the Fukushima Plant?
On April 6, the radiation level in the city of Fukushima was 13.9 μSv/h. If we multiply that number by the number of hours in a year, the yearly radiation dose is about 122,000 μSv. This number is more concerning. Remember, the U.S. does not allow workers to receive more than 50,000 μSv per year at work.
We do know that several Fukushima staff members and firefighters have been exposed to dangerous levels of radiation. At least six staffers were exposed to radiation levels greater than 100,000 μSv (potentially increasing their risk of cancer). At least 50 firefighters received doses of 27,000 μSv.
So How Does Fukushima Compare with Chernobyl?
The level of radiation detected at the Fukushima plant immediately after the third explosion was 400,000 μSv/h–a level that would be extraordinarily dangerous for humans. By comparison, that level of radiation is 33,000 times higher than we recorded about 750 yards from the containment facility at reactor 4 in Chernobyl. Of course, Chernobyl happened 25 years ago and has a cement sarcophagus over it.
It is still too early to speculate on the lasting dangers created by the disaster at the Fukushima plant. Like Chernobyl, it is likely that an exclusionary zone will be created around the plant, and that some residents who lived nearby will never return home.
If you would like to learn more about radiation risks and scientific disputes on the issue, check out the following New York Times article titled Radiation Is Everywhere, But How To Rate Harm.
Again, we recommend watching the documentary The Battle of Chernobyl. It details the incredible circumstances that followed the Chernobyl accident, and the unbelievable sacrifices made to prevent a much larger disaster.
And Finally, More Photos from the Chernobyl Exclusion Zone
Click here to see the complete virtual tour.









This post is from Drew McCartor, Pure Earth’s Director of Global Policy and Planning. It was published in 2011.
Learn more about Pure Earth’s projects to address pollution.






Your readings are way off and low.
Here’s the current data from Fukushima
Live: 196 locations
Futaba, Fukushima, Japan
Median reading in this area is approximately
328.000 µSv per hour ( 2873280.000 µSv per year )
4049.38272x the average public space geiger reading for Japan (0.081 µSv per hour)*
328.000% the radiation dose you would receive from a chest x-ray (100 µSv)*
Tag: sensor:type=radiation
Current reading: 328.000 µSv per hour Time: 2011-07-14 13:06:3
You can see all the current data here: http://japan.failedrobot.com/
Also Tepco has a live webcam here: http://mfile.akamai.com/127380/live/reflector:51361.asx?bkup=52045&prop=a
Hi Ron,
Thank you for your comment and for providing a link to new radiation data. This blog post was written in April and therefore does not represent current radiation levels. We are pleased that you have added additional sources of information, and we invite you and others to continue to comment and highlight new resources. Thanks.
you make a statement which I dispute
You state:
“10 μSv – The average radiation you received today”
maybe if I had an XRay or worked in a dangerously radioactive place … which I do not ..
10 microSv per day = over .4 microSv/hr
= approx 4milliSv per year
= four times the common legal maximum exposure for adult male (non nuke worker)
= more than ten times the common recommended maximum exposure for pregnant women and infants
Better call it “Radiation 911”
What is natural radiation and WHAT IS MAN MADE RADIATION.
This is what you totally ignore.
And what ARE dose limits? You do not answer this.
Man made radionuclides like
Cesium 137 – mimics potassium
Strontium 90 – mimics Calcium
Iodine 131 – mimics Idodine
dit NOT exist before atomic age.
Natural Radiation is mostly gamma and electro magnetic, along with Radium, Radon, Uranium, Tritium (mostly atmosphere, mimics hydrogen, thanks to reactors inscreases millions of times), and so on.
But THIS is Full list of MAN MADE radionuclides:
http://www.life-upgrade.com/DATA/inventory-reactor-beznau2.jpg
Trillions and Trillions of Becquerel. Only ONE Reactor.
In 1970 it was proven that human fetus is 500 times more vulnerable to radiation. And in 1978 this was released in the bulletin of atomic scientists: http://books.google.de/books?id=aAoAAAAAMBAJ&pg=PA30&lpg=PA30&dq=cancer+stewart+xray+1970+radiographs&source=bl&ots=UGZYt0TZGo&sig=ENE9wYZjjNs3Rh2XyptdZwP3Ucw&hl=de&ei=7545Tu6iF8aAOsLrvbMG&sa=X&oi=book_result&ct=result&resnum=1&sqi=2&ved=0CBsQ6AEwAA#v=onepage&q&f=false
ICRP has confirmed in 1966 that natural radiation is harmful! ICRP Publication Number 8 from 1966 on page 60. in relation to the health damage caused by natural radiation for the bulk of the world’s population is a risk of sixth order (1 to 10 dead per million per rad / gray) in a few areas with high natural background radiation the risk fifth order. 10 to 100 dead per million and rad (gray). and in 1977 the ICRP publication number 26 said, that, in this sense, regional differences of the natural radiation are so regarded, that the corresponding differences include the damage. and in the ’80s the natural radiation was simply doubled. and in 2011 it became the twentyfold in japan: 20 mSv/a. Natural radiation is used as an excuse to increase artificial radiation with radionuclide-antagonists.
They call that radiation-protection!
1971: the discoverer of plutonium and president of the USAEC Glenn Theodore Seaborg resigns – at the same time the ICRP reduced the additional maximum dose near reactors for the public from 500 mrem/annual (5 mSv/a) to 5 mrem/annual (0,05 mSv/a).
the USAEC “protects” health and advertises nuclear industry at the same time, later renamed in AEC, then renamed in NRC.
Source NRC (USAEC / AEC before): http://connection.ebscohost.com/c/articles/32111590/seaborg-resigns-from-aec
Source (ICRP 5 mSv/a): http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2596042/pdf/yjbm00117-0064.pdf
! no coincidence !
Thanks to Prof. Ernest Sternglass and his studies about infant mortality near reactors;: http://www.ratical.org/radiation/inetSeries/ejs1192.html
And, in 1991, the ICRP lowered the standard from 5 mSv /a to 1 mSv/a: “For occupational exposure in planned exposure situations the Commission now recommends an equivalent dose limit for the lens of the eye of 20 mSv in a year, averaged over defined periods of 5 years, with no single year exceeding 50 mSv.” page 1: Source: http://www.icrp.org/docs/ICRP%20Statement%20on%20Tissue%20Reactions.pdf
And in 2011, the japanese government increased it for infants, students, mature, unborn, to: 20 mSv/a
A 2000 PERCENT INCREASE.
Source: http://www.heraldsun.com.au/news/breaking-news/japanese-ire-over-radiation-limit-for-kids/story-e6frf7jx-1226061484710
20 mSv mean: below, but still high: 555,000 becquerel per m² contamination (from 1995 Belarus National Report): http://tekknorg.files.wordpress.com/2012/02/162.jpg
17 milli Sievert per year mean: 16,000 – 32,000 additional cancer deaths AND 150,000 – 1,5 million deaths by genetic diseases: http://www.ratical.org/radiation/CNR/PP/app1.html by John W. Gofman, Ph.D., M.D and Arthur R. Tamplin, Ph.D. quote “If we use the most optimistic Russell mouse genetic data, and even if we give full credit for slow delivery of radiation, we reach the conclusion that 100,000 extra genetic deaths per year would occur for the allowable average exposure of 170 millirads to the population. This can hardly be construed as an “optimistic” outlook, or a “safe” dose of radiation.”
http://tekknorg.wordpress.com/2011/03/19/effect-of-cesium-and-strontium-on-japanese-children-japanese-officals-irresponsible/
NUCLEAR HOLOCAUST in SLOW MOTION.
“…To provide an adequate safety standard the dose limit of 1 mSv/y have to be reduced to 0.02 mSv/y or 20 µSv/y.”Page 9: http://www.staff.uni-marburg.de/~kunih/all-doc/stoakuni.pdf by Dr. Horst Kuni, Nuclear Medicine, University Professor.
Professor Yuri Bandazhevsky, a pathologist, Rector of the Medical Institute of Gomel, on the ingestion of radio caesium includes – he said: “Clinical checks on children between 1996 and 1999 show that at levels greater than 50Bq/kg there are pathological changes in vital organs and systems – cardiovascular, nervous, endocrine, immune, reproductive, digestive excretory and eyes. Caesium concentrations in the placenta reveal a relationship with nervous system defects in the foetus. The health condition of the population is a disaster but being a physician myself I cannot accept it as hopeless. With all my faith in God and life I appeal to anyone who can influence it: do your best to improve the situation. There is nothing more precious on this planet than life. And we should do everything possible to protect it.” http://www.spokesmanbooks.com/Spokesman/PDF/91Gifford.pdf
Until 1990 ICRP said it is not necessary to evacuate people, as long as the radiation does not exceed 500 mSv.: http://books.google.de/books?id=Ber3ENERfGwC&pg=PA343&lpg=PA343&dq=ICRP+40++500mSv&source=bl&ots=IaOqT2MqK2&sig=l3l0MYGe_nKkaXxvFZxkkG8hP5M&hl=de&ei=gc81TpqyI4PfsgajxLG5Ag&sa=X&oi=book_result&ct=result&resnum=8&ved=0CE0Q6AEwBw#v=onepage&q=ot%20exceed%20500%20mSv.%20The%20ICRP&f=false
WARNING TO ALL PREGNANT WOMEN: http://tekknorg.wordpress.com/2011/08/03/safe-radiation-levels-never-get-your-facts-here/
IAEA subsequently took its radiation protection recommendations directly from ICRP (rather than WHO), therefore persons from the Commission who also sit on UNSCEAR both make the rules and judge their adequacy: http://www.ratical.org/radiation/Chernobyl/CaUFtH.html
From every Reactor Chminey (according to General Electric): Every second: 0,01 Curie + 50 micro curie = 371,850,000 decays. Decay can mean: Tritium transmutates into Helium 3. And it mimics hydrogen. Where hydrogen is (body / food / DNA) there can be tritium. Whether the decay times are short, does not matter (iodine 131). Because every single radionuclid has a chance to get collected in the soil. And is constantly released by the chimney. The soil is programmed to collect. There our fruits, plants, vegetables grow (eaten by us and animals). The radionculides from reactors can be transported up to 400 km and more. The affinity factors are for example Growth time and depth of the roots.
“Radiation can reduce antioxidant levels dramatically because of the use of antioxidants to eliminate free radicals produced in the presence of radiation.” PETKAU EFFECT: “The production of free radicals of Oxygen (O² with a negative electric charge) caused by ionising effect of the radiation. The free radicals caused by the slow dose (0,001) are sparsely distributed radicals do have a higher chance to reach the cell membranes.” http://onlinelibrary.wiley.com/doi/10.1111/j.1365-2664.2007.01353.x/abstract;jsessionid=90C315EEFA3E5B978EFA17BBB75F03EB.d02t02#.T0fbqeWicyA.facebook
The principle of the slow motion nuclear holocaust: “Weakening of the victim, then another radiation shocks” -> Based on the long term follow up of the population exposed to ionizing radiation due to residence near nuclear testing fields in the period 1949-1962 increased chromosome aberration were detected not only in directly exposed population but also in their children and grandchildren. Unstable chromosome aberrations ( dicentrics and rings) were detected in children of parents who were exposed to radiation as part of anticancer therapy. Cells of such children also express higher radiosensitivity. http://www.springerlink.com/content/f373917263h7hxg6/
If the water is 1000 times above normal, the organisms in it are many 100,000 times of times above normal. http://life-upgrade.com/DATA/FUKUSHIMA-SEA-BECQUEREL.jpg Full Decay Time Cesium 137: 301 years
with kind regards,
And for a true insight, I recommend this movie, in parts, to you and every reader:
Nuclear Controversies 1 – 4
#1 http://www.youtube.com/watch?v=8qqhm_ZrfhE
#2 http://www.youtube.com/watch?v=LqHjfyT5Dmk
#3 http://www.youtube.com/watch?v=1T4kezJEcpY
#4 http://www.youtube.com/watch?v=cIv6eIE8Ljo
The Sacrifice 1 – 2
# 1 http://www.youtube.com/watch?v=lNc4MvI2s08
#2 http://www.youtube.com/watch?v=PA3_PRjHxuM
70 – 90% comes from internal radiation. FOOD, SOIL.
Not AIR. Sievert has little meaning.
Refer to becquerel and gamma spektrometer / whole body counters.
All radiation dose limits we have today are based on “T65D” study on Hiroshima and Nagasaki = Counting ONLY external radation effects
withd kind regards,
Hello,
The article is quite simple and informative. Thank you for this nice post!
Could you also add some insights on how the intensity of radiation spreads over the distance (with similar numerical examples)? I read somewhere it follows the distance-squared law.